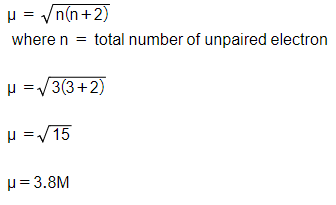ReasonThe spin only magnetic moment of an ion is equal to sqrt {n(n+2)} where n is the number of unpaired electrons in the ion.AssertionThe spin only magnetic moment of Sc^{3+} is 1.73
![The spin-only magnetic moment value for the complex [Co(CN6)]4- is ______ BM. [At. no. of Co = 27] - Chemistry (JEE Main) | Shaalaa.com The spin-only magnetic moment value for the complex [Co(CN6)]4- is ______ BM. [At. no. of Co = 27] - Chemistry (JEE Main) | Shaalaa.com](https://www.shaalaa.com/images/_4:7a962dcc0b6e460d9cf2e9f2567e4097.png)
The spin-only magnetic moment value for the complex [Co(CN6)]4- is ______ BM. [At. no. of Co = 27] - Chemistry (JEE Main) | Shaalaa.com
![The complex with spin only magnetic moment of ~4.9 BM is:a)[Fe(H2O)6]2+b)[Fe(CN)6]3-c)[Fe(CN)6]4-d)[Fe(H2O)6]3+Correct answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question The complex with spin only magnetic moment of ~4.9 BM is:a)[Fe(H2O)6]2+b)[Fe(CN)6]3-c)[Fe(CN)6]4-d)[Fe(H2O)6]3+Correct answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question](https://edurev.gumlet.io/ApplicationImages/Temp/9569037_ab072f64-2e6d-4920-b550-ecfc86b0a8de_lg.png)
The complex with spin only magnetic moment of ~4.9 BM is:a)[Fe(H2O)6]2+b)[Fe(CN)6]3-c)[Fe(CN)6]4-d)[Fe(H2O)6]3+Correct answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question
What will be the theoretical value of spin only magnetic field when Fe(SCN)3 reacts with the solution containing F ions to yield a complex

SOLVED: 'The calculated spin only magnetic moment for Ti2+ metal ion is a 3.87 b 1,.73 C. 4.92 d. 2.83'

No links please Calculate spin only magnetic moment of Fe3+ ion - Chemistry - Alcohols Phenols and Ethers - 12302421 | Meritnation.com







![Spin only magnetic moment of the compound Hg [C0 (SCN)(4)] is Spin only magnetic moment of the compound Hg [C0 (SCN)(4)] is](https://static.doubtnut.com/ss/web/416446.webp)


![ANSWERED] 140 The calculated spin only magnetic moment of Cr ion is 1 - Kunduz ANSWERED] 140 The calculated spin only magnetic moment of Cr ion is 1 - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20201001105030189875-2167239.jpg)